
| Drug | Paclitaxel |
| Coating | Polymer Free Abluminal |
| Dosage | 2.5 µg/mm2 |
| Material | Cobalt Chromium Alloy L605 |
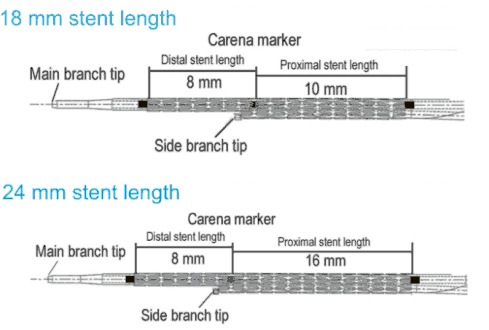
| Length | 18 and 24 mm |
| Diameter | See Table in “Product references” |
| Number of cells | 7/8/6 or 9/10/8 |
| Metal to artery ratio | From 10 to 15% |
| Stent thickness | 73 µm (+5 µm drug coating) |
| Nominal pressure | 8 atm |
| Rated Burst Pressure | 14 atm |
| Guidewire compatibility | 0.014” |
| Guiding catheter compatibility | 6F |
| DIAMETER (mm) | LENGTH (mm) | ||
| Main branch balloon (mm) | Side branch balloon (mm) | 18mm | 24mm |
| 2.5 | 2.0 | NIPAX001 | NIPAX0011 |
| 3.0 | 2.0 | NIPAX002 | NIPAX012 |
| 3.0 | 2.5 | NIPAX003 | NIPAX013 |
| 3.5 | 2.5 | NIPAX004 | NIPAX014 |
| 3.5 | 3.0 | NIPAX005 | NIPAX015 |
| 2.5 | 2.5 | NIPAX006 | NIPAX016 |
| 3.0 | 3.0 | NIPAX007 | NIPAX017 |

Minvasys quality system is ISO 9001 and ISO 13485 certified, hence giving full support to its international partners for local market authorizations. Minvasys devices comply with the requirements of the European Medical Devices Directive 93/42/CEE.
Minvasys
7, rue du Fossé Blanc - 92230 Gennevilliers - France
Tel : +33 (0) 1 47 90 70 30
Fax : +33 (0) 1 47 91 05 85
info@minvasys.com