
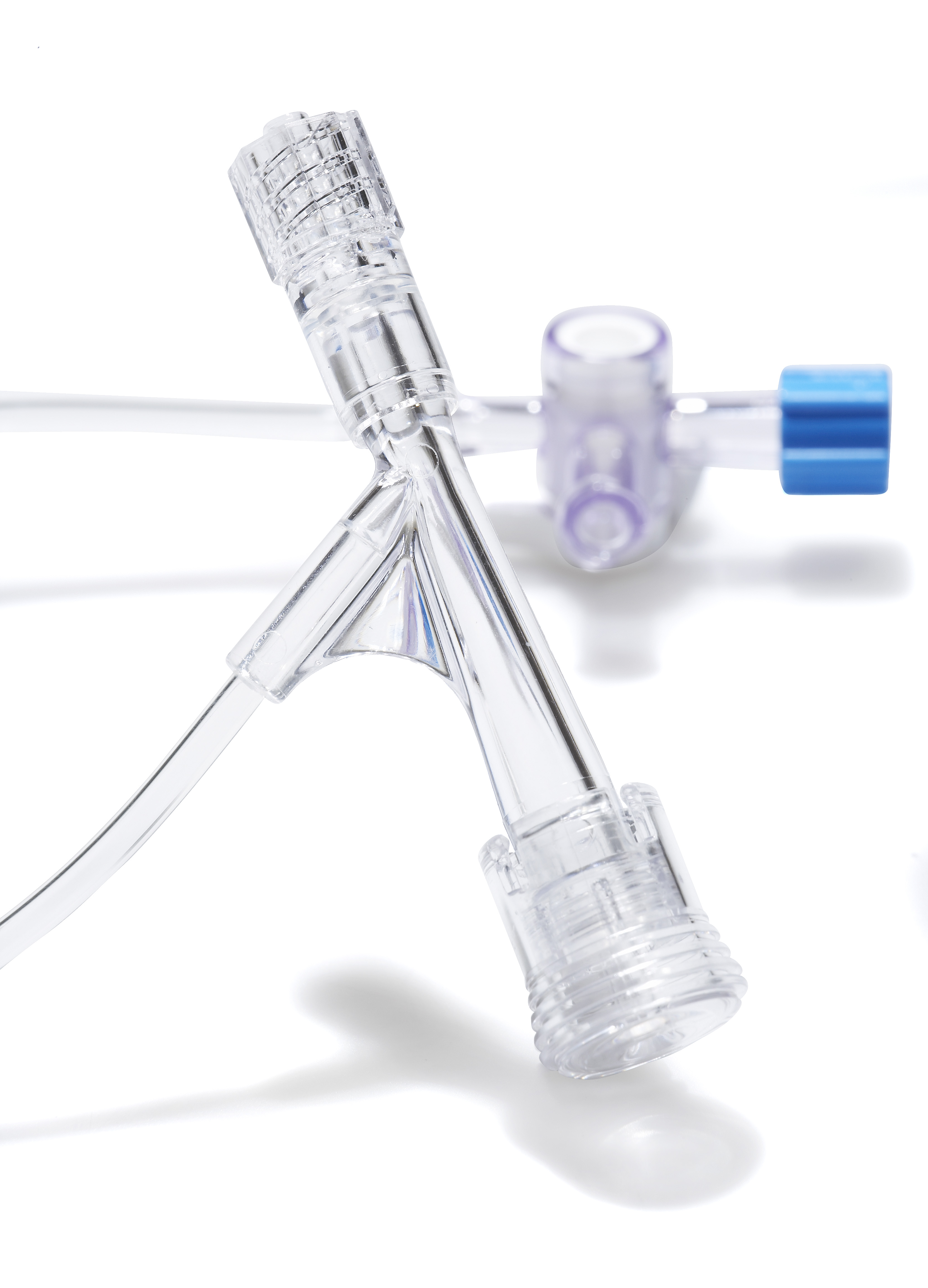
| Compatibility with devices to be inserted (max) | 7F / 0.092” / 2.33 mm |
| Metallic insertion tool internal diameter | 1.9F / 0.025” / 0.64 mm |
| Plastic peelable insertion tool (Speed Ketch only): claimed microcatheter compatibility (max) | Up to 3F |
| Torquer (Super Ketch Plus, Super Ketch LSA and Speed Ketch Plus only): guide wire compatibility (min/max) | 0.010’’ to 0.035’’ |
| REFERENCES | |||||
| SPEEDKETCH | SUPERKETCH | SUPERKETCH LSA | TORQUER | INSERTION TOOL | |
| KESU0001 | 20cm side arm | Up to 0.025" | |||
| KESU0002 | 20 cm side arm | From 0.010" to 0.035" | Up to 0.025" | ||
| KESU0003 | 50cm side arm | Up to 0.025" | |||
| KESU0004 | No side arm | Up to 0.025" | |||
| KESU0005 | 50cm side arm | From 0.010" to 0.035" | Up to 0.025" | ||
| KESU0006 | No side arm | From 0.010" to 0.035" | Up to 0.025" |

Minvasys quality system is ISO 9001 and ISO 13485 certified, hence giving full support to its international partners for local market authorizations. Minvasys devices comply with the requirements of the European Medical Devices Directive 93/42/CEE.
Minvasys
7, rue du Fossé Blanc - 92230 Gennevilliers - France
Tel : +33 (0) 1 47 90 70 30
Fax : +33 (0) 1 47 91 05 85
info@minvasys.com